April 2019
Rita Mourão Rosa, Lisa Alexandra Pereira Mestrinho
Original article: Acute phase proteins in cats
ABSTRACT: Acute phase proteins (APP) are proteins synthesized and released mainly by hepatocytes when cells are damaged or invaded by microorganisms. This article reviews the use of APPs in feline diseases, identifies their utility in the clinical setting, and analyzes 55 published papers. Serum amyloid A (SAA), alpha-1 acid glycoprotein (AGP), and haptoglobin are markers that the authors consider useful in monitoring the acute inflammatory response in cats. Although measurement of APP is still not routinely used in veterinary medicine, together with clinical signs and other blood parameters, they are of clinical interest and applicable in diseases such as feline infectious peritonitis, pancreatitis, renal failure, retroviral and calicivirus infections. Although there are commercially available kits for measuring feline APP, standardization of assays aimed at technical simplicity, greater species specificity, and lower associated costs will enable routine use in feline practice, as is done in the human field.
keywords: inflammation, acute phase proteins, cat.
Introduction
Acute phase response (APR) is an early nonspecific systemic innate immune response to a local or systemic stimulus that helps treat and restore homeostasis and minimize tissue damage when an organism is affected by trauma, infection, stress, surgery, neoplasia, or inflammation (GRUYS et al. , 2005; CRAY et al., 2009; ECKERSALL AND BELL, 2010). In this reaction, we observe several different systemic effects: fever, leukocytosis, hormonal changes - mainly cortisol and thyroxine concentrations, with secondary catabolic status and serum muscle, iron and zinc depletion (CERÓN et al. 2005, JAVARD et al. 2017).
Cytokines IL-1β, TNF-α, and especially IL-6, and approximately 90 minutes after injury, increase protein synthesis in hepatocytes, lymph nodes, tonsils, and spleen, as well as blood leukocytes. These newly formed proteins are called acute phase proteins (APPs) (TIZARD, 2013b).
Acute-phase proteins
APP concentrations may increase (APP positive) or decrease (APP negative) in response to inflammation (PALTRINIERI et al., 2008) (JOHNSTON & TOBIAS, 2018). They can activate leukocytosis and complement, cause protease inhibition, lead to blood clotting and opsonization - a defense mechanism that leads to the elimination of infectious agents, tissue regeneration and restoration of health (CRAY et al., 2009). APP can have two functions, pro- and / or anti-inflammatory, which must be fine-tuned to promote homeostasis (HOCHEPIED et al., 2003).
According to the size and duration of the reaction following the stimulus, three main groups of APP are distinguished (MURATA et al., 2004; PETERSEN et al., 2004; CERÓN et al.). Positive APP can be divided into two groups: the first group includes APP with an increase of 10 up to 1000-fold in humans or 10- to 100-fold in domestic animals in the presence of inflammation - e.g. c-reactive protein (CRP) and serum amyloid A (SAA). The second group are APPs, which increase 2 to 10-fold in an inflammatory response - e.g. haptoglobin and alpha-globulins. The last group included negative APP, in which the concentration decreases in response to inflammation - e.g. albumin (KANN et al., 2012).
Acute phase positive proteins
Positive APPs are glycoproteins whose serum concentrations, when stimulated by pro-inflammatory cytokines, increase by 25 % during the disease process and are released into the bloodstream. These concentrations can be measured and used in diagnosis, prognosis, monitoring of response to treatment, as well as general health screening. They can also be considered as quantitative biomarkers of the disease, highly sensitive to inflammation but not very specific, as an increase in APP can also occur in non-inflammatory diseases (CERÓN et al., 2005; ECKERSALL and BELL, 2010).
Positive APPs respond to cytokines differently, and these groups fall into two main classes. Type 1 APP, which includes AGP, complement component 3, SAA, CRP, haptoglobin and hemopexin, is regulated by IL-1, IL-6 and TNF-α as well as glucocorticoids. Type 2, which includes three fibrinogen chains (α-, β- and γ-fibrinogen) and various inhibitory proteases, is regulated by cytokines IL-6 and glucocorticoids (BAUMANN et al., 1990; BAUMANN & GAULDIE, 1994).
In cats, the most important APP is SAA (Serum Amyloid A) or alpha-1-acid glycoprotein (AGP). The level of SAA in the blood can indicate inflammatory conditions such as feline infectious peritonitis (FIP) and other infectious diseases such as calicivirus infection, chlamydiosis, leukemia and infectious immunodeficiency, as it increases 10- to 50-fold (TIZARD, 2013b). SAA can also be elevated in other diseases such as diabetes mellitus and cancer. Haptoglobin is usually increased 2- to 10-fold and is particularly high in FIP (TIZARD, 2013b). Table 1 summarizes the individual positive APPs in the context of feline disease.
Acute phase negative proteins
The most significant negative APP is albumin, whose blood concentration decreases during APR due to amino acid aberrations towards the synthesis of positive APPs (CRAY et al., 2009; PALTRINIERI, 2007a). Other negative APPs are transferrin, transthyretin, retinol ligand, and cortisol binding protein, proteins involved in vitamin and hormone transport (JAIN et al., 2011).
Acute phase proteins in cat disease
Unlike cytokines, which are small in size and rapidly filtered by the kidney, acute phase proteins have a higher molecular weight (greater than 45 kDa) and consequently remain in plasma for longer (SALGADO et al., 2011).
APP levels can only indicate inflammation, and consequently their concentrations can help diagnose and monitor the disease. APP can help detect subclinical inflammation, distinguish acute from chronic disease, and predict its course (VILHENA et al, 2018; JAVARD et al., 2017). Because APRs begin before specific immunological changes occur, they can be used as an early marker of disease before leukogram changes occur, with their magnitude related to disease severity (PETERSEN et al., 2004; CÉRON et al., 2005; VILHENA et al., 2005). , 2018). For this reason, disease monitoring can be considered one of the most interesting and promising applications of APP.
APP levels along with clinical signs and blood tests have been evaluated in a variety of animal diseases (ie, FIP, canine inflammatory disease, leishmaniasis, ehrlichiosis, and canine pyometra) and have been shown to be useful in diagnosis, response to treatment, and prognosis (ECKERSALL et al. ), 2001; MARTINEZ-SUBIELA et al., 2005; SHIMADA et al., 2002; JERGENS et al., 2003; GIORDANO et al., 2004; PETERSEN et al., 2004; DABROWSKI et al., 2009; VILHENA et al., 2018).
To obtain complete information on APR, one major and one moderate positive as well as one negative APP should be evaluated simultaneously (CERÓN et al., 2008). High concentrations of major APP are usually associated with infectious diseases, usually systemic bacterial infection or immune-mediated disease (CERÓN et al., 2008; TROÌA et al., 2017). Although APPs should be analyzed along with white blood cell and neutrophil counts, they are most sensitive in the early detection of inflammation and infection (CERÓN et al., 2008; ALVES et al., 2010). However, the specificity of these proteins is low in determining the cause of the process, and also increases in physiological conditions such as pregnancy (PALTRINIERI et al., 2008).
| APP | The disease |
|---|---|
| SAA | FIP Induced inflammation and surgery Various diseases (pancreatitis, renal failure, FLUTD, tumors, diabetes mellitus; kidney disease, injury, etc.) Sepsis FeLV; hemotropic mycoplasma infections Hepatozoonfelis and Babesia vogeli infection Dirofilariaimmitis FIV cats treated with recombinant feline interferon |
| AGP | Chlamydophila psittaci infection; Pancreatitis and pancreatic tumors FIP Lymphoma and other tumors Induced inflammation and surgery FIV cats treated with recombinant feline interferon Abscesses, pyothorax, adipose tissue necrosis Various diseases (FLUTD, tumors, diabetes mellitus, kidney diseases, injuries, etc.) |
| Haptoglobin | FIP Induced inflammation and surgery Abscesses, pyothorax, adipose tissue necrosis Various diseases (FLUTD, tumors, diabetes mellitus, kidney diseases, injuries, etc.) Hepatozoonfelis and Babesia vogeli infection FeLV, hemotropic mycoplasmas Dirofilariaimmitis |
| CRP | FIV cats treated with recombinant feline interferon Induced inflammation and surgery |
Legend: Serum amyloid A (SAA), α1-acid glycoprotein (AGP), systemic inflammatory response syndrome (SIRS), feline lower urinary tract disease (FLUTD), feline infectious peritonitis (FIP), feline leukemia virus (FeLV), immunodeficiency virus cats (FIV); feline calicivirus (FCV).
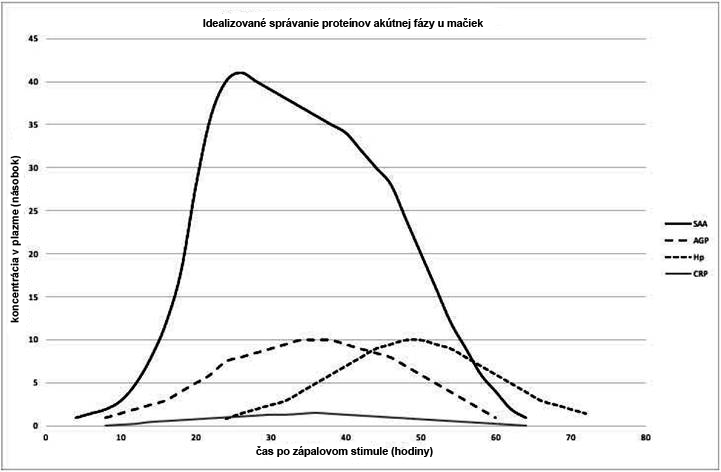
Figure 1 shows the expected behavior of acute phase positive proteins based on revised studies. AGP, SAA and haptoglobin have been identified as useful indicators for monitoring the acute inflammatory response in cats (WINKEL et al., 2015; PALTRINIERI et al., 2007a, b; KAJIKAWA et al., 1999). APPs in cats were first identified after comparative measurements in the serum of clinically normal and diseased animals, in experimentally induced inflammation studies, and in postoperative studies (KAJIKAWA et al., 1999). The concentration of SAA reportedly increased first, followed by an increase in AGP and haptoglobin, in contrast to a less pronounced increase in CRP (KAJIKAWA et al., 1999). One study showed that CRP behaves similarly to SAA and AGP in cat inflammation (LEAL et al., 2014).
Serum Amyloid A
SAA is one of the major APPs in several species, important in both humans and cats (KAJIKAWA et al., 1999). It modulates the immune response by attracting inflammatory cells to tissues and leading to the production of multiple inflammatory cytokines (GRUYS et al., 2005; TIZARD, 2013a). Its concentration can increase more than 1,000 times in an inflammatory condition, which we then understand as inflammation (TAMAMOTO et al., 2013). However, such an increase can be observed in both non-inflammatory and inflammatory diseases and neoplasms (TAMAMOTO et al., 2013). According to a study in cats that underwent surgery, SAA levels begin to increase approximately 3 to 6 hours, peaking 21 to 24 hours after surgery (SASAKI et al., 2003).
Alpha 1-acid glycoprotein
Alpha 1-acid glycoprotein (AGP) is an acute phase-reactive protein found in the serum mucoid portion of serum (SELTING et al., 2000; WINKEL et al., 2015). Like most positive APPs, AGP is a glycoprotein synthesized predominantly by hepatocytes in APR and released into the bloodstream (CÉRON et al., 2005).
AGP can be used to monitor early interferon treatment in cats infected with feline immunodeficiency virus (FIV) (GIL et al., 2014). AGP as well as haptoglobin (Hp) are increased in anemic cats suffering from pyothorax, abscesses or fat necrosis (OTTENJANN et al., 2006).
Changes in AGP in feline neoplasia do not appear to be consistent across studies. Some of them do not describe any changes in cats with lymphoma (CORREA et al., 2001). Others point to an increase in both AGP and SAA in cats with sarcomas, carcinomas, or other round cell tumors (SELTING et al., 2000; TAMAMOTO et al., 2013; MEACHEN et al., 2015; HAZUCHOVA et al., 2017).
AGP is important as an indicator test for FIP, which is used specifically in Europe (CECILIANI et al., 2004). GIORI et al. examined the specificity and sensitivity of several tests in 12 cats, with 33.33 % cats being FIP negative based on histopathology and immunohistochemistry and 66.66 % cats being FIP positive confirmed by histopathology and immunohistochemistry. This author concludes that immunohistochemistry must always be performed to confirm FIP, but high concentrations of AGP can help support the diagnosis of FIP if immunohistochemistry cannot be performed and histopathology is not convincing.
Haptoglobin
Haptoglobin (Hp) is one of the most important acute phase proteins in cattle, sheep, goats, horses and cats (TIZARD, 2013a), synthesized mainly by hepatocytes but also by other tissues such as skin, lungs and kidneys (JAIN et al, 2011 ). Hp binds to iron molecules and makes them inaccessible to invasive bacteria, thereby inhibiting bacterial proliferation and invasion. Subsequently, it also binds to free hemoglobin, thus preventing its oxidation with lipids and proteins (TIZARD, 2013a), which justifies a reduction in Hp in case of hemolysis.
In cats, Hp usually increases 2- to 10-fold in inflammatory conditions, and is particularly high in FIP (TIZARD, 2013a). However, both Hp and SAA did not provide sufficient support to distinguish FIP from other causes of effusion compared to AGP (HAZUCHOVÁ et al., 2017).
Measurement APP
The serum is composed of a large number of individual proteins in which the detection of changes in its fractions can provide important diagnostic information (ECKERSALL, 2008).
Ideally, measurement of all serum proteins should be available so that they can be used as a diagnostic tool in relation to inflammatory diseases.
Currently, APPs (Table 2) can be determined by enzyme-linked immunosorbent assay (ELISA), radioimmunoassay, nephelometry, immunoturbidimetry (IT), Western blot, and messenger ribonucleic acid (mRNA) analysis (CÉRON et al., 2005; PALTRINIERI et al., 2008; SCHREIBER et al., 1989). Although some human APP tests have been automated for veterinary medicine, species-specific tests are still limited. Cross-species differences in APP and the limited availability of cross-reactive agents have so far contributed to the low routine level of APP determination in veterinary laboratories, especially in cats. Regardless, the technology is evolving and routine monitoring of clinically relevant APPs in cats can be expected in the near future.
Conclusion
Acute phase proteins in cats are biomarkers suitable for monitoring inflammation, along with other clinical and laboratory findings that are useful in diagnosing subclinical changes, monitoring the development and effect of the disease in the body, as well as in evaluating the response to treatment.
In cats, SAA APP, which is most pronounced in response to inflammation, is followed by AGP and haptoglobin, in contrast to CRP, which is used in other species.
Although there are commercially available kits for determining feline APPs, standardization of tests for technical simplicity, higher species specificity with lower associated costs will allow routine use in feline practice, as is done in human medicine.
| Analyzes | Pros | Cons |
|---|---|---|
| Radioimmunoassay | 24 to 48 hours to obtain results, specific operator skills required | |
| ELISA | Commercially available species-specific kits | Lack of automation, expensive, some "between-run" inaccuracy |
| Immunoturbidimetry | 30 minutes to obtain results, customizable with a biochemical analyzer | |
| Western Blot | Long time for immunoblot processing | |
| Nephelometric immunoassays | They depend on the cross-reactivity of the increased antiserum |
Appendix: APP and their position in the electrophoretogram
Although there are tests directly for a specific APP, it is useful to know in which region the electrophoretograms are located.

| Serum protein | Electrophoretic region |
|---|---|
| α1-acid glycoprotein | α1 (alpha-1) |
| Serum Amyloid A | α (alpha) |
| Haptoglobin | α2 (alpha-2) |
| Ceruloplasmin | α2 (alpha-2) |
| Transferrin | β1 (beta-1) |
| C-reactive protein | γ (gamma) |
References
- ALVES, AE et al. Leucogram and serum acute phase protein concentrations in queens submitted to conventional or videolaparoscopic ovariectomy. Arquivo Brasileiro de Medicina Veterina- ria e Zootecnia, v.62, n.1, p.86-91, 2010. Available from:. Accessed: Oct. 10, 2018. doi: 10.1590 / S0102-09352010000100012.
- BAUMANN, H. & GAULDIE, J. The acute phase response.
Immunol Today, v.15, n.2, p.74-80, 1994. Available from:
https://doi.org/10.1016/0167-5699(94)90137-6. Accessed: Aug. 21, 2018. doi: 10.1016 / 0167-5699 (94) 90137-6. - BAUMANN, H. et al. Distinct regulation of the interleukin-1 and interleukin-6 response elements of the rat haptoglobin gene in rat and human hepatoma cells. Molecular and Cellular Biology, v.10, n.11, p.5967–5976, 1990. Available from: Accessed: Aug. 21, 2018. doi: 10.1128 / MCB.10.11.5967.
- BENCE, L. et al. An immunoturbidimetric assay for rapid quantitative measurement of feline alpha-1-acid glycoprotein in serum and peritoneal fluid. Veterinary Clinical Pathology, v.34, n.4, p335-341, 2005. Available from:. Accessed: Jan. 13, 2019. doi: 10.1111 / j.1939-165X.2005.tb00058.x.
- CALLAHAN, G. & YATES, R. Veterinary Clinical Laboratory Immunology. In Warren, A. Basic Veterinary Immunology, pp. 295-317, 2014. Boulder, Colorado: University Press of Colorado.
- CECILIANI, F. et al. Decreased sialylation of the acute phase protein α1-acid glycoprotein in feline infectious peritonitis (FIP). Veterinary Immunology and Immunopathology, v.99, n.3- 4, p.229-236, 2004. Available from:. Accessed: Aug. 24, 2018. doi: 10.1016 / j. vetimm.2004.02.003.
- CERON, J. et al. Acute phase proteins in dogs and cats: current
knowledge and future perspectives. Veterinary Clinical - Pathology, v.34, n.2, p.85-99, 2005. Available from:. Accessed: Aug. 20, 2018. doi: 10.1111 / j.1939-165X.2005.tb00019.x.
- CERÓN, JJ A seven-point plan for acute phase protein interpretation in companion animals. Veterinary Journal, v.177, n.1, p.6-7, 2008. Available from:. Accessed: Aug. 20, 2018. doi: 10.1016 / j. tvjl.2007.12.001.
- CORREA, SS et al. Serum alpha 1-acid glycoprotein concentration in cats with lymphoma. Journal of the American Animal Hospital Association, v.37, n.2, p.153-158, 2001. Available from:
https://doi.org/10.5326/15473317-37-2-153. Accessed: Aug. 24, 2018. doi: 10.5326 / 15473317-37-2-153. - CRAY, C. et al. AcutePhase Response in Animals: A Review. Comparative Medicine, v.59, n.6, p.517–526, 2009. Available from:. Accessed: Aug. 21, 2018.
- DABROWSKI, R. et al. Usefulness of C-reactive protein, serum amyloid A component and haptoglobin determinations in bitches with pyometra for monitoring early postovariohysterectomy complications. Theriogenology, v.72, n.4, p.471–476, 2009. Available from:. Accessed: Aug. 23, 2018. doi: 10.1016 / j.theriogenology.2009.03.017.
- DUTHIE, S. et al. Value of α1-acid glycoprotein in the diagnosis of feline infectious peritonitis. The Veterinary Record, v.141, n.12, p.299–303, 1997. Available from:. Accessed: Aug. 11, 2018. doi: 10.1136 / vr.141.12.299.
- ECKERSALL, P. Proteins, Proteomics, and the Dysproteinemias. In Kaneko, J., Harvey, J. & Bruss, M. In Clinical Biochemistry of Domestic Animals. 6th ed. USA: Elsevier, 2008, Chap. 5, pp.117-155.
- ECKERSALL, PD & BELL, R. Acute phase proteins: Biomarkers of infection and inflammation in veterinary medicine. The Veterinary Journal, v.185, n.1, p.23-27, 2010. Available from:. Accessed: Aug. 20, 2018. doi: 10.1016 / j.tvjl.2010.04.009.
- ECKERSALL, PD et al. Acute phase protein response in an experimental model of ovine caseous lymphadenitis. BMC Veterinary Research, v.19, p.3-35, 2007. Available from:. Accessed: Aug. 24, 2018. doi: 10.1016 / j.tvjl.2010.04.009.
- ECKERSALL, PD et al. Acute phase proteins in serum and milk from dairy cows with clinical mastitis. Veterinary Record, v.148, n.2, p.35–41, 2001. Available from:. Accessed: Aug. 22, 2018. doi: 10.1136 / vr.148.2.35.
- GIL, S. et al. Oral recombinant feline interferon-omega as an alternative immune modulation therapy in FIV positive cats: Clinical and laboratory evaluation. Research in Veterinary Science, v.96, n.1, p.79–85, 2014. Available from:. Accessed: Oct. 10, 2018. doi: 10.1016 / j.rvsc.2013.11.007.
- GIORDANO, A. et al. Changes in some acute phase protein and immunoglobulin concentrations in cats affected by feline infectious peritonitis or exposed to feline coronavirus infection. The Veterinary Journal, v.167, n.1, p.38-44, 2004. Available from:
https://doi.org/10.1016/S1090-0233(03)00055-8. Accessed:
Aug. 9, 2018. doi: 10.1016 / S1090-0233 (03) 00055-8. - GIORI, L. et al. Performances of different diagnostic tests for feline infectious peritonitis in challenging clinical cases. Journal of Small Animal Practice, v.52, n.3, p.152-157, 2011. Available from:
https://doi.org/10.1111/j.1748-5827.2011.01042.x. Accessed:
Aug. 24, 2018. doi: 10.1111 / j.1748-5827.2011.01042.x. - GRUYS, E. et al. Acute phase reaction and acute phase proteins. Journal of Zhejiang University. Science B, v.6, n.11, p.1045- 1056, 2005. Available from:. Accessed: Aug. 21, 2018.
doi: 10.1631 / jzus.2005.B1045. - HAZUCHOVA, K. et al. Usefulness of acute phase proteins in differentiating between feline infectious peritonitis and other diseases in cats with body cavity effusions. Journal of Feline Medicine and Surgery, v.19, n.8, p.809-816, 2017. Available from: https://doi.org/10.1177/1098612X16658925. Accessed: Aug. 11, 2018. doi: 10.1177 / 1098612X16658925.
- HOCHEPIED, T. et al. α1-Acid glycoprotein: an acute phase protein with inflammatory and immunomodulating properties. Cytokine Growth Factor Rev, v.14, n.1, p.25–34, 2003. Available from:
https://doi.org/10.1016/S1359-6101(02)00054-0. Accessed: Aug. 21, 2018. doi: 10.1016 / S1359-6101 (02) 00054-0. - JACOBSEN, S. et al. Evaluation of a commercially available human serum amyloid A (SAA) turbidometric immunoassay for determination of equine SAA concentrations. Veterinary Journal, v.172, n.2, p.315–319, 2006. Available from:. Accessed: Aug. 24, 2018. doi: 10.1016 / j.tvjl.2005.04.021.
- JAIN, S. et al. Acute-phase proteins: As diagnostic tool. Journal of Pharmacy and Bioallied Sciences, v.3 v.1, p.118–127, 2011. Available from: https://www.ncbi.nlm.nih.gov/pmc/articles/PMC3053509/. Accessed: Aug. 21, 2018. doi: 10.4103 / 0975-7406.76489.
- JAVARD R. et al. Acute phase proteins and iron status in cats with chronic kidney Disease. Journal of Veterinary Internal Medicine, v.31, n.2, p.457-464, 2017. Available from:. Accessed: Oct. 10, 2018. doi: 10.1111 / jvim.14661.
- JERGENS, AE et al. A scoring index for disease activity in canine inflammatory bowel disease. Journal of Veterinary Internal Medicine, v.17, n.3, p.291–297, 2003. Available from:. Accessed: Aug. 22, 2018. doi: 10.1111 / j.1939-1676.2003.tb02450.x.
- KAJIKAWA, T. et al. Changes in concentrations of serum amyloid A protein, alpha 1-acid glycoprotein, haptoglobin, and C-reactive protein in feline sera due to induced inflammation and surgery. Veterinary Immunology and Immunopathology, v.68, n.1, p. 91-98, 1999. Available from: Accessed: Aug. 10, 2018. doi: 10.1016/S0165-2427(99)00012-4.
- KANN, R. et al. Acute phase proteins in healthy and sick cats. Research in Veterinay Science, v.93, n.2. p.649-654, 2012. Available from: https://doi.org/10.1016/j.rvsc.2011.11.007. Accessed: Aug. 20, 2018. doi: 10.1016 / j.rvsc.2011.11.007.
- KURIBAYASHI, T. et al. Alpha 1-acid glycoprotein (AAG) levels
in healthy and pregnant beagle dogs. Experimental Animals, v.52,
n. 5, p.377–381, 2003. Available from:. Accessed: Jan. 13, 2019. doi: 10.1538 / expanim.52.377. - LEAL, R. et al. Monitoring acute phase proteins in retrovirus infected cats undergoing feline interferon-ω therapy. Journal of Small Animal Practice, v.55, n.1, p.39-45, 2014. Available from:
https://doi.org/10.1111/jsap.12160. Accessed: Jan. 6, 2019. doi: 10.1111 / jsap.12160. - MARTÍNEZ-SUBIELA, S. et al. Analytical validation of commercial techniques for haptoglobin determination, C reactive protein and amiloid A series in canines [Analytical validation of commercial techniques for haptoglobin, C reactive protein and serum amyloid A determinations in dogs]. Archivos de Medicina Veterinaria, v.37, n.1, 2005. Available from:. Accessed: Jan. 13, 2019. doi: 10.4067 / S0301-732X2005000100009.
- MEACHEM, MD et al. A comparative proteomic study of plasma in feline pancreatitis and pancreatic carcinoma using 2-dimensional gel electrophoresis to identify diagnostic biomarkers: A pilot study. Canadian Journal of Veterinary Research, v.79, n.3, p.184-189, 2015. Available from:. Accessed: Oct. 10, 2018.
- MURATA, H. et al. Current research on acute phase proteins in veterinary diagnosis: An overview. The Veterinary Journal, v.168, n.1, p.28–40, 2004. Available from:. Accessed: Aug. 20, 2018. doi: 10.1016 / S1090-0233 (03) 00119-9.
- OTTENJANN, M. et al. Characterization of the anemia of inflammatory disease in cats with abscesses, pyothorax, or fat necrosis. Journal of Veterinary Internal Medicine, v.2, n.5, p. 1143-1150, 2006. Available from:. Accessed: Aug. 24, 2018. doi: 10.1111 / j.1939-1676.2006.tb00713.x.
- PALTRINIERI, S. Early biomarkers of inflammation in dogs and cats: The acute phase protein. Veterinary Research Communications, v.31, n.1, p.125-129, 2007a. Available from:
. Accessed: Aug. 21, 2018. doi: 10.1007 / s11259-007-0107-3. - PALTRINIERI, S. et al. Serum alpha1-acid glycoprotein (AGP) concentration in non-symptomatic cats with feline coronavirus (FCoV) infection. Journal of Feline Medicine and Surgery, v.9, n.4, p.271-277, 2007b. Available from:. Accessed: Aug. 11, 2018. doi: 10.1016 / j. jfms.2007.01.002.
- PALTRINIERI, S. The feline acute phase reaction. Review. The Veterinary Journal, v.111, n.1, p.26-35, 2008. Available from:
https://doi.org/10.1016/j.tvjl.2007.06.005. Accessed: Aug. 24, 2018. doi: 10.1016 / j.tvjl.2007.06.005. - PETERSEN, H. et al. Application of acute phase protein measurements in veterinary clinical chemistry. Veterinary Research, v.35, n.2, p.163–187, 2004. Available from:. Accessed: Aug. 20, 2018. doi: 10.1051 / vetres: 2004002.
- SALGADO, FJ, et al. (2011). Acute phase proteins as biomarkers of disease: from Bench to Clinical Practice. In Veas, F. Acute Phase Proteins as Early Non-Specific Biomarkers of Human and Veterinary Diseases. Rijeka, Croatia: InTech. Available from:
http://www.documentation.ird.fr/hor/fdi:010060045. Accessed:
Aug. 21, 2018. doi: 10.5772 / 1045. - SASAKI, K. et al. Evaluation of feline serum amyloid A (SAA) as an inflammatory marker. Journal of Veterinary Medical Science, v.65, n.4, p.545-8, 2003. Available from:. Accessed: Aug. 10, 2018.
- SCHREIBER, G. et al. The acute phase response in the rodent. Annals of the New York Academy of Science, v.557, p.61–85, 1989. Available from:. Accessed: Aug. 24, 2018. doi: 10.1111 / j.1749- 6632.1989.tb24000.x.
- SELTING, K. et al. Serum alpha 1-acid glycoprotein concentrations in healthy and tumor-bearing cats. Journal of Veterinary Internal Medicine, v.14, n.5, p.503-506, 2000. Available from:. Accessed: Aug. 9, 2018. doi: 10.1111 / j.1939-1676.2000.tb02267.x.
- SHIMADA, T. et al. Monitoring C-reactive protein in beagle dogs experimentally inoculated with Ehrlichiacanis. Veterinary Research Communications, v.26, n.3, p.171–177, 2002. Available from:. Accessed: Aug. 22, 2018. doi:
10.1023 / A: 1015290903332. - SILVESTRE-FERREIRA, AC et al. Serum acute phase proteins in Dirofilariaimmitis and Wolbachia seropositive cats. Journal of Feline Medicine and Surgery, v.19, n.6, p.693–696, 2017. Available from: https://doi.org/10.1177/1098612X15625435. Accessed: Sep. 16, 2018. doi: 10.1177 / 1098612X15625435.
- TAMAMOTO, T. et al. Serum amyloid A as a prognostic marker in cats with various diseases. Journal of Veterinary Diagnostic Investigation, v.25, n.3, p.428–432, 2013. Available from:. Accessed: Jan. 27, 2019.
- TECLES, F. et al. Validation of a commercially available human immunoturbidimetric assay for haptoglobin determination in canine serum samples. Veterinary Research Communications, v.31, n.1, p.23–36, 2007. Available from:. Accessed: Jan. 13, 2019. doi: 10.1007 / s11259-006-3397-y.
- TERWEE, J. et al. Characterization of the systemic disease and ocular signs induced by experimental infection with Chlamydia psittaci in cats. Veterinary Microbiology, v.59,
259-281, 1998. Available from:. Accessed: Aug. 20, 2018. doi: 10.1016 / S0378-1135 (97) 00185-5. - TIZARD, I. Innate immunity: proinflammatory and antimicrobial mediators / systemic responses to inflammation. In Veterinary Immunology. 9.ed. St. Louis, Missouri: Saunders, Elsevier, 2013a. Chap. 6, pp.52-58.
- TIZARD, I. Innate immunity: proinflammatory and Atimicrobial mediators / systemic responses to Inflammation. In Veterinary Immunology. 9.ed. St. Louis, Missouri: Saunders, Elsevier, 2013b. Chap. 4, pp.31-40.
- TROÌA, R. et al. Serum amyloid A in the diagnosis of feline sepsis. Journal of Veterinary Diagnostic Investigation, v.29, n.6, p.856-859, 2017. Available from:. Accessed: Aug. 11, 2018. doi: 10.1177 / 1040638717722815.
- VILHENA, H. et al. Acute phase proteins response in cats naturally infected by hemotropic mycoplasmas. Comparative Immunology, Microbiology & Infectious Diseases, v.56, p.1-5, 2018. Available from: https://doi.org/10.1016/j.cimid.2017.11.001. Accessed: Aug. 11, 2018. doi: 10.1016 / j.cimid.2017.11.001.
- VILHENA, H. et al. Acute phase proteins response in cats naturally infected with Hepatozoonfelis and Babesia vogeli. Veterinary Clinical Pathology, v.48, n.1, p.72-76, 2017. Available from:
https://doi.org/10.1111/vcp.12451. Accessed: Aug. 10, 2018. doi: 10.1111 / vcp.12451. - WEIDMEYER, C. & SOLTER, P. Validation of human haptoglobin immunoturbidimetric detection of haptoglobin in equine and canine serum and plasma. Veterinary Clinical Pathology, v.24, n.4, p.141–146, 1996. Available from:. Accessed: Jan. 13, 2019. doi: 10.1111 / j.1939-165X.1996.tb00988.x.
- WHICHER, T. et al. Immunonephelometric and immunoturbidimetric assays for proteins. Critical Reviews in Clinical Laboratory Sciences, v.18, n.3, p.213–260, 1983. Available from:
https://doi.org/10.3109/10408368209085072. Accessed: Aug. 13, 2019. doi: 10.3109 / 10408368209085072. - WINKEL, V. et al. Serum α-1 acid glycoprotein and serum amyloid A concentrations in cats receiving antineoplastic treatment for lymphoma. American Journal Veterinary Research, n.76, v.11, p.983-988, 2015. Available from:. Accessed: Aug. 22, 2018. doi: 10.2460 / ajvr.76.11.983.


